Researchers are one step closer to finding a cure for one type of inflammatory bowel disease (IBD). A group of microbial enzymes living in the gut microbiome were found to be the culprit behind ulcerative colitis.
Ulcerative colitis is a chronic illness affecting the colon of nearly million people living in the United States. The current findings not only find the cause behind overactive inflammation but also shows a potential treatment route for blocking it.
“Studies continue to show correlations between gut health and microbial constituents, but these trends don’t exactly explain how the bacteria cause disease or what we can do about it,” says study co-senior author David J. Gonzalez, PhD, associate professor of pharmacology at UC San Diego School of Medicine and Skaggs School of Pharmacy and Pharmaceutical Sciences in a media release. “This is the first study with experimental evidence that pinpoints a specific microbe driving ulcerative colitis, the protein class it expresses, and a promising solution.”
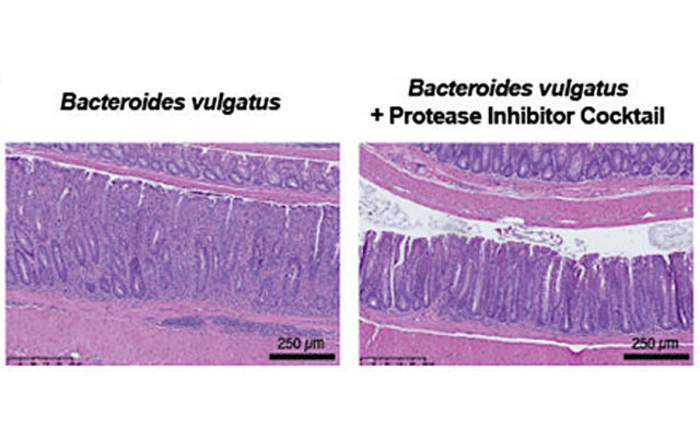
The researchers used stool samples to study bacteria that originated from the gut microbiome. It’s a useful and less invasive method compared to traditional blood or tissue samples. Almost 40% of patients with ulcerative colitis had high amounts of proteases — enzymes that break down other proteins — that come from the gut bacterial species Bacteroides vulgatus.
Blocking protein degradation showed to be an effective way of alleviating ulcerative colitis symptoms. In an animal study, researchers transplanted human feces containing a high number of proteases into mice. Mice given the protease-rich feces acquired ulcerative colitis. However, protease inhibitors significantly decreased ulcerative colitis symptoms.
Based on the findings, the team hypothesizes that the increase in bacterial protease production may come from a stressor in the gut, such as nutrient deprivation, that forces the body to use protein as an alternative nutrient source. However, too much proteases may damage the lining of the colon. Colon damage may then trigger immune cells to cluster and produce high levels of inflammation.
The study was recently published in the journal Nature Microbiology.
