A recent mouse study suggests a reason behind colitis is the activation of a receptor that promotes inflammation and pain. Blocking the protease-activated receptor could be a target for stopping high inflammation that irritates the colon.
The digestive system contains a high number of enzymes that break down proteins called proteases. These enzymes are made from multiple sources, including the gut microbiome, inflammatory cells, or digestive enzymes in the intestines.
While proteases help in digesting proteins from food, they also signal cells by activating specific G protein-coupled receptors (GCPR). GCPRs are a family of receptors regulating many bodily processes and are the target for one-third of drugs. When proteases activate a GCPR–protease-activated receptor-2 (PAR2) on nerve cells, it signals cells involved in producing pain.
Previous research has associated protease activation of PAR2 in many gastrointestinal diseases that cause pain including inflammatory bowel disease, irritable bowel syndrome, and cancer. But scientists poorly understood the mechanism behind colon pain.
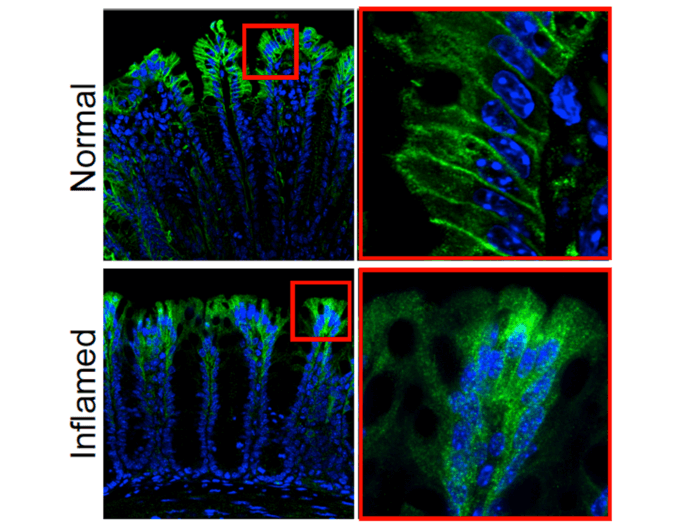
The current study used genetically modified mice where PAR2 is tied to a green fluorescent protein to make it more visible when there is a cell that expresses PAR2. Results showed the cells that expressed PAR2 were in the surface or membrane of epithelial cells that line the small and large intestines. Some were also expressed in the nerve fibers of these areas.
In healthy mice, they found PAR2 on the membrane of colonic epithelial cells. However, in mice with colitis, cells expressing PAR2 shifted from the surface of cells to compartments in cells called endosomes. Moving into endosomes created signals that eventually lead to inflammation and pain because it disrupts the normal protective function of cells lining the colon.
“We identified not only where this receptor is in the digestive tract, but also how it signals inflammation and pain in the colon,” says senior author Nigel Bunnett, PhD, professor and chair of the Department of Molecular Pathobiology at NYU College of Dentistry, in an NYU release. “This more complete understanding of PAR2 and its signaling mechanism could ultimately help us to better treat inflammatory and painful diseases of the colon.”
Additional studies with human colon tissue confirmed that activating PAR2 produced inflammation in the colon.
When researchers blocked the movement of the PAR2 receptor from entering cells by stopping the expression of a protein called dynamin-2 it stopped signaling of cells and, more importantly, significantly decreased pain and inflammation.
“This could be achieved through blocking PAR2 from entering cells, as we did in this study by inhibiting dynamin-2,” says Dr. Bunnett. “It could also mean getting drugs that activate PAR2 not just to the surface of cells, but into the interior of cells using nanoparticles to reach the receptor in endosomes,” says Dr. Bunnett.
The study is published in the Proceedings of the National Academy of Sciences (PNAS).