Antibiotic treatment can be, and frequently is, lifesaving. Unfortunately, antibiotics not only kill the bacteria causing an infection, but they also indiscriminately kill or disturb the balance of the trillions of bacteria making up a healthy human microbiota. The result may be as benign as transient diarrhea or it can be life-threatening. A team of scientists at Harvard University and the Massachusetts Institute of Technology (MIT) collaborated to develop a live biotherapeutic product (eLBP) that, when given together with commonly used antibiotics, protects the gut microbiome from adverse disturbance.
The antibiotics with the most frequent harmful effects are beta-lactams, including penicillin. The term for the alteration in the gut microbiota is “dysbiosis.” It triggers short-term diarrhea in up to 35 percent of patients. It can take months to resolve. Often, the diarrhea requires additional treatment. In some patients the microbiota can be permanently disturbed, becoming a serious risk factor for a host of autoimmune, metabolic, and neurological diseases.
“In designing the eLBP, we tapped into the synthetic biology took kit that we have advanced over the past two decades and enabled Lactococcus lactis, a safe-to-use microbe, to secrete a b-lactamase enzyme that degrades b-lactams in the bacteria’s environment,” says James Collins, Ph.D. core faculty and lead of the Institute’s Living Cellular Device platform, in a statement. “The enzyme essentially cannot confer a selective advantage to the producing bacteria or be easily transferred to other bacteria, minimizing the risk of adverse effects and maximizing the clinical benefits of our approach.”
Usually, b-lactamase enzymes are encoded by just one gene. These are passed between bacteria via a process called horizontal gene transfer. The enzymes are in the bacterial cell wall or membrane enclosures. This not only makes the producing bacterial strain resistant to certain antibiotics that attack outer enclosures, but also allows their resistance to spread to other bacteria.
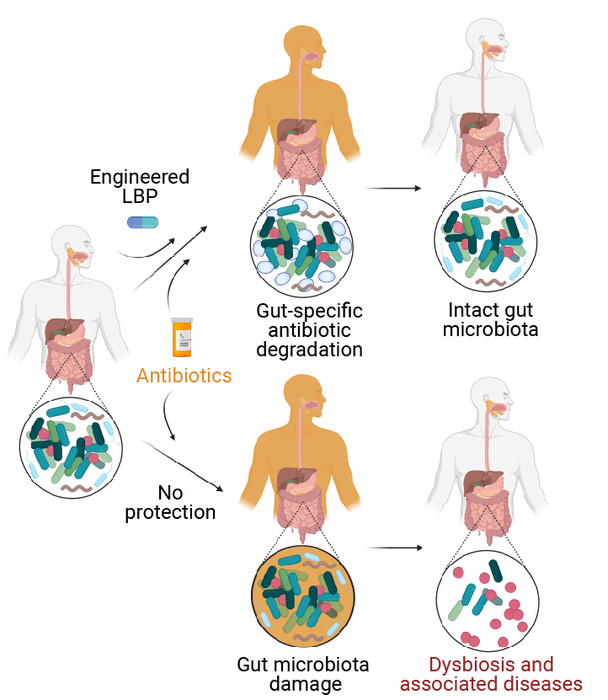
Cubillos-Ruiz and his co-workers on Collins’ team then showed that when they gave their eLBP intervention to mice that received the antibiotic ampicillin as an oral treatment, it minimized dysbiosis in each animal’s gut. They found that the eLBP significantly lessened the adverse alteration of microbial populations and allowed them to fully recover their original diversity and composition three days after antibiotic treatment. Mice treated with ampicillin unprotected by the eLPB sustained a much greater loss of their microbial diversity which could not be recovered.
“Importantly, during its transient stay in the digestive tract, the eLBP protected the microbiome without changing the concentration of circulating ampicillin. Maintaining the concentration is important because the antibiotic still needs to reach infections everywhere else in the body to do its job,” says Cubillos-Ruiz.
“The eLBP also reduced the enrichment of various antibiotic resistance genes within the microbial community, which commonly happens under the selective pressure of antibiotics,” he continues.
The gut microbiome contains a natural pool of bacteria with genes, including b-lactamase genes, that induce resistance to antibiotics through different mechanisms – including resistance even to unrelated antibiotics.
Next, the team addressed a frequent consequence of dysbiosis: takeover of the intestinal territory by problematic bacteria like Clostridium difficile. These occupy the intestines at lower numbers, but when given the chance to multiply uncontrollably, they trigger inflammation, diarrhea, and possibly inflammatory bowel disease. The eLBP prevents the intestinal colonization with problematic bacteria.
“We are now focusing on getting these living therapies to patients and are finalizing the design of an effective, brief, and inexpensive clinical trial,” says Cubillos-Ruiz, adding that, “We also believe that our general eLBP approach can be extended into a therapeutic platform that could be applied not only to other antibiotics, but also to address diseases where gut dysbiosis is at the center.”
“This elegantly engineered and highly effective living cellular therapeutic device could become a true game changer in the treatment of infectious diseases both by helping to maintain a healthy microbiome in patients treated with antibiotics and, perhaps equally important in the longer run, by preventing antibiotic resistance which is a growing problem worldwide,” says Wyss Founding Director Dr. Donald Ingber.
The study is published in Nature Biomedical Engineering.
